|
8/18/2023 0 Comments Gold element atomic number Magnesia, a district of Eastern Thessaly in GreeceĪlumina, from Latin alumen (gen. Symbol Na is derived from Neo-Latin natrium, coined from German Natron, ' natron'.Greek elements hydro- and -gen, ' water-forming'īeryl, a mineral (ultimately from the name of Belur in southern India) īorax, a mineral (from Arabic bawraq, Middle Persian * bōrag) For more detailed information about the origins of element names, see List of chemical element name etymologies. Like the periodic table, the list below organizes the elements by the number of protons in their atoms it can also be organized by other properties, such as atomic weight, density, and electronegativity. It is a tabular arrangement of the elements by their chemical properties that usually uses abbreviated chemical symbols in place of full element names, but the linear list format presented here is also useful. The definitive visualisation of all 118 elements is the periodic table of the elements, whose history along the principles of the periodic law was one of the founding developments of modern chemistry. A chemical element, often simply called an element, is a type of atom which has the same number of protons in its atomic nucleus (i.e., the same atomic number, or Z). This is a list of the 118 chemical elements that have been identified as of 2023. Bismuth telluride has been used in thermoelectric devices.List of the 118 identified chemical elements Tellurium is used as a basic ingredient in blasting caps, and is added to cast iron for chill control. Tellurium improves the machinability of copper and stainless steel, and its addition to lead decreases the corrosive action of sulfuric acid on lead and improves its strength and hardness. Natural tellurium consists of eight isotopes. Thirty isotopes of tellurium are known, with atomic masses ranging from 108 to 137. Workmen exposed to as little as 0.01 mg/m 3 of air, or less, develop "tellurium breath," which has a garlic-like odor. 
Tellurium and its compounds are probably toxic and should be handled with care. Molten tellurium corrodes iron, copper, and stainless steel. 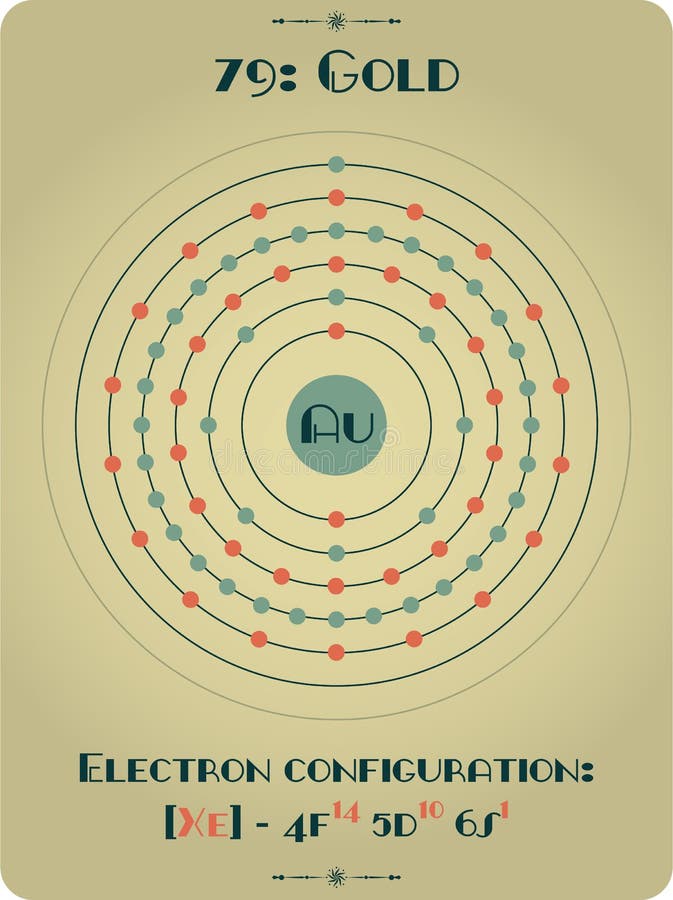
In air, tellurium burns with a greenish-blue flames, forming the dioxide. It can be doped with silver, copper, gold, tin, or other elements. Its conductivity increases slightly with exposure to light. Tellurium is a p-type semiconductor, and shows greater conductivity in certain directions, depending on alignment of the atoms. Whether this form is truly amorphous, or made of minute crystals, is open to question. Gold Facts Gold is the only metal that is yellow or 'golden. This is the only truly yellow metal on Earth, but there's a lot more to learn about gold. Amorphous tellurium is found by precipitating tellurium from a solution of telluric or tellurous acid. Updated on JanuThere are many interesting facts about the element gold, which is listed on the periodic table as Au. PropertiesĬrystalline tellurium has a silvery-white appearance, and when pure it exhibits a metallic luster. 
The U.S., Canada, Peru, and Japan are the largest Free World producers of the element. 
It is recovered commercially from anode muds produced during the electrolytic refining of blister copper. Tellurium is occasionally found native, but is more often found as the telluride of gold (calaverite), and combined with other metals. Discovered by Muller von Reichenstein in 1782 named by Klaproth, who isolated it in 1798. Back to Elements List Tellurium Atomic Number:Ħ, 5, 4, 3, 2, 1, −1, −2 (a mildly acidic oxide)įrom the Latin word tellus, earth. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
